
The largest sources of CO2 emissions are carbon-containing fuel-fired power production and the transport sector. To reduce a major part of emissions, CO2 can be captured from thermal power plants in parallel with the production of hydrogen; subsequently, hydrogen can be an energy carrier for the transport sector.
Hydrogen is an attractive alternative as an energy carrier, avoiding the two biggest problems associated with carbon-containing fuels - limited reserves, and CO2 emissions.
Hydrogen can be produced industrially by hydrocarbon reforming (or coal gasification), or by water electrolysis. In water electrolysis, water molecules are split into hydrogen and oxygen by electrical energy:
Reaction: 2H2O + electric energy →2H2 + O2
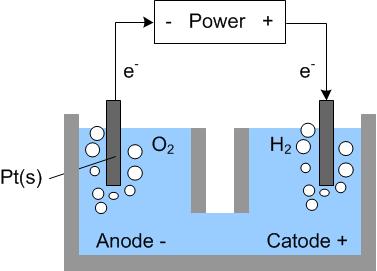
| Anode reaction: | 2H2O → O2 + 4H+ + 4e- | - ε˚ = - 1.23 V |
| Cathode reaction: | 4H2O + 4e-→ 2H2 + 4OH- | ε˚ = - 0.83 V |
| Net reaction: | 6H2O → 2H2 + O2 + 4(H+ + OH-) | |
| 2H2O → 2H2 + O2 | ε˚ = - 2.06 V |
Hydrogen production from hydrocarbon reforming contributes to CO2 emissions if the by-product, CO2, is not captured and stored in a suitable underground structure. Absorption would be a suitable technology to separate H2 from CO2 where typically 80 – 90% of the CO2 produced can be captured.
The figure below shows the process solution that integrates an oxyfuel power process with hydrogen production from water electrolysis. Coal is used as an example of a carbon-containing fuel. The oxyfuel process supplies electric power and water to the water electrolysis and the water electrolysis supplies oxygen to the oxyfuel power process. This invention is groundbreaking because of the integration of water electrolysis and the oxyfuel power process.
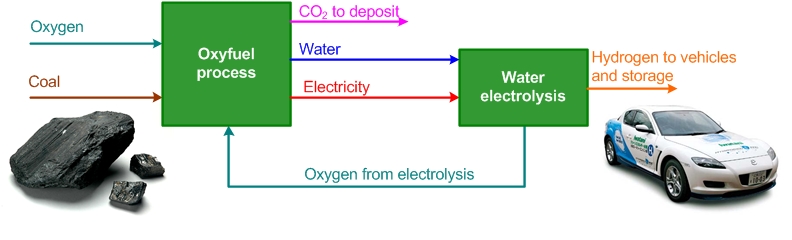
The advantages of the integrated process solution are:
Nebb provide total solutions for automation, electrical and information systems to a number of industries world-wide, including process industry, food & pharma, marine, energy, subsea, and PMC.
Our core competence lies within automation, electrical, instrumentation, SCADA, software, and digitalisation.
[fa icon="phone"] +47 66908300
[fa icon="envelope"] sales@nebb.com
[fa icon="envelope"] general@nebb.com
[fa icon="home"] Solbråveien 43, Asker Norway